By Karen Harrington
EDITOR’S SUMMARY: Cancer rates are rising—and one of the biggest drivers isn’t in your water, your food or your genes. It’s your lights. You’ll see how after-dark illumination undermines hormonal timing, accelerates tumor growth and reshapes cancer patterns across continents. The takeaway is clear: improving your light environment may be one of the most powerful cancer-prevention steps you can take.
In the modern world, cancer touches many people’s lives. As you think about the recent rise in cancer rates—affecting both the young and old—you may likely consider the role of chemical and heavy metal pollutants, microplastics and pesticides. You may also question shifting lifestyles, the rise of obesity or exposure to pharmaceuticals. While all of these factors are important, there is another major player that is almost always missed in the lineup—even by those committed to living a healthy, anti-cancer lifestyle: after-dark artificial lighting. And it’s not just fluorescent bulbs but also LEDs and even incandescents.
The idea seems absurd in a world where electric lighting is simply part of the backdrop of daily life. Light has long been viewed as something that keeps your nights illuminated. And yet the light bulb itself is used as a symbol of innovation. But light bulbs—especially those emitting broad-spectrum light—come with a hidden cost. When light bulbs emit white light, which has a blue light component, and are turned on after dark, they produce light at night (LAN) exposure. LAN significantly disrupts hormone balance and has emerged as a major contributor to cancers that are driven by hormone imbalance—primarily breast and prostate cancers.
It may not be obvious that visible light affects reproductive hormones and the cancers linked to them. Yet a relationship between light exposure and hormone release runs throughout the animal kingdom. This connection is exploited heavily by chicken farmers, who carefully manage the lighting environment in hen houses to control the rates of sexual maturation of hens and maximize egg laying: “Light stimulation (usually an increase of as little as one hour) has an immediate effect on the production of reproductive hormones,” as noted in the Hy-Line technical guide “The Effect of Light on the Reproductive System of Poultry.”
The biological reason for this relationship is the need for animals in many environments to achieve pregnancy and give birth at the appropriate season. The duration of daylight is used by the body to help determine what season it is, and thus what level of reproductive hormones should be released. The intermediary between light exposure and reproductive hormone release is the multitasking super hormone melatonin, better known for its role in helping you sleep at night. The pineal gland produces melatonin when it senses an absence of blue light, although green wavelengths and overall brightness can also have some effect. Even species that no longer reproduce seasonally—including humans—still rely on melatonin to help regulate their reproductive hormones. When the total duration of daily blue-light exposure is artificially extended by electric lighting, however, hormonal signaling becomes disrupted.
The propensity of modern humans to be indoors during the day, missing out on daytime sunshine, is another important factor. While melatonin is produced in darkness, its precursors are made with the help of the sun. Morning sunlight also signals the body to increase melatonin levels at night. Thus optimal hormonal regulation by melatonin requires both adequate darkness exposure during the night/winter, and sufficient outdoor sunlight exposure during the day/summer.
Electrification, Menarche, and the Global Rise of Hormone-Driven Cancers
The introduction of electric lighting to human populations started in the late 1800s. First it impacted streetlighting, factories and big city businesses. Then it moved into the homes of the wealthy. Soon it spread to upscale neighborhoods. But by 1910, the price of providing electricity had dropped low enough to start providing electric lighting to the average urban resident. Over the ensuing three decades, nearly everyone across the developed world received home electricity and came to regard it as an indispensable part of their daily lives.
The effect of electric lighting on reproductive hormones was apparent as early as 1910, but the cause and effect went unrecognized and remains overlooked even today, despite published research. The first sign was a sudden drop in the age at menarche—when girls typically get their first period. The pattern repeated across countries and cultures as electric lighting was introduced to different parts of the world. A particularly stark data set exists for India, where electrification moved gradually through part of the country during most of the twentieth century and then reached the rest of the country very quickly as the national grid was built between 1994 and 2018. The figure below is from Pathak et al. (2014). Note that it is plotted by year of birth, so the first clear drop appears in girls born in the early to mid 1980s; they reached puberty in the mid 1990s just as the national grid was going in.

In parts of sub-Sahara Africa that do not have widespread electricity today, the average age of menarche is still 14 ½, matching what was seen throughout the world before industrialization, even though these girls are subject to other forms of modern pollution. In addition, exposure to electric light at night can also impact fertility. That’s because the same melatonin signals that guide puberty also shape ovulation and other reproductive processes. For post-menopausal women, undue disruption of melatonin can affect estrogen in ways that lead to obesity. For nursing women, melatonin levels in breast milk change with the time of day, so improper light exposure can disrupt those levels and interfere with the baby’s circadian rhythm entrainment. And then there’s where this all leads: the hormonally driven cancers.
If you have not personally experienced breast or prostate cancer, chances are that someone close to you has. American women and men currently have a one-in-eight chance of developing one of these cancers in their lifetimes. Globally, breast cancer is now the most commonly diagnosed cancer in the world, accounting for approximately 11.7% of all new cancer cases in 2020. The International Agency for Research on Cancer (IARC) classifies several well-established breast cancer–related exposures as Group 1 carcinogens. According to IARC classifications, alcoholic beverages, combined estrogen–progestogen contraceptives, diethylstilbestrol (DES), and exposure to X- or γ-radiation are all confirmed human carcinogens with documented links to breast cancer.
Taken together, these factors explain approximately 50% of breast cancer cases, making it clear that something else significant is also going on as cancer rates rise. Shift work—because it exposes you to mistimed light and chronic circadian disruption—has been identified as a prime suspect, and now carries a Class 2A (probably carcinogenic) designation from the IARC. That 2A classification was reaffirmed in 2019 as additional evidence accumulated linking circadian disruption with breast cancer. More broadly, compelling research has been emerging for decades showing that the issue extends beyond the intense nighttime illumination faced by shift workers. It appears that everyday after-dark lighting among all residents of electrified regions may account for the missing 50%. You can think about it this way…electric lighting nudges your body into a shift-worker pattern, as your bedtime naturally drifts later when after-dark light is present.
A large body of laboratory and clinical evidence shows that melatonin helps suppress breast-cancer growth, and that blue-light exposure at night interferes with this protective effect. In one novel experiment—“Melatonin-Depleted Blood from Light-Exposed Humans Promotes Tumor Growth in Human Breast Cancer Xenografts”—researchers grafted human breast cancer onto lab rats and then injected the rats with blood that was either melatonin-depleted (taken from women who had worked the night shift) or melatonin-enriched (from women who had avoided nighttime light exposure). The rats receiving the melatonin-enriched blood showed tumor regression, while tumors continued to grow in those given melatonin-depleted blood.
In one observational study titled “Total Visual Blindness Is Protective Against Breast Cancer,” researchers found that women with no light perception had markedly lower breast-cancer rates than blind women who could still detect light, highlighting the role of light sensing in melatonin disruption. Broader population data point in the same direction: a 2021 meta-analysis of 17 cohort and case-control studies found that higher levels of indoor or outdoor light-at-night exposure were associated with increased breast-cancer risk overall, with stronger associations in pre-menopausal women. Together, these findings reinforce the importance of a truly dark nighttime environment in maintaining stable melatonin rhythms.

Latitude, Light Pollution, and Cancer Latency
Breast and prostate cancer rates rise with increasing latitude, where daylight hours are shorter and seasonal light patterns are more extreme. Yet before electrification, even at the highest latitudes, Indigenous Arctic populations showed almost no breast or prostate cancer, despite having documented cases of other cancers such as lung and salivary gland cancers. The latitude pattern may stem from the suppression of melatonin during the long summer days at high latitude. In theory, this should be balanced by extra melatonin production during the long, dark winter evenings—but evening electric lighting prevents this seasonal recovery. As a result, the substantial melatonin loss in high-latitude winters under electric lights likely overwhelms the protective benefits of maintaining a dark bedroom during sleep.
In fact, the amount of breast and prostate cancer caused by evening exposure to electric light before bedtime is extremely difficult to pin down because, unlike light during sleeping hours, nearly everyone in developed nations is exposed to artificial illumination before bed. Thus it is necessary to look at people living in undeveloped areas and try to account for other variables. Kloog et al. (2010) did this exercise and found a 30–50 percent difference in breast cancer rates between countries with the most and least light pollution, after adjusting for known confounders.
Historically, before the introduction of electric lighting, breast cancer was exceedingly rare—although most other types of cancer were too, at least outside of cities. In urban areas from dynastic Egypt to medieval Europe, people often spent long hours indoors surrounded by smoke and soot, with limited exposure to the sunshine needed to produce melatonin precursors. Under these conditions, cancer—including breast cancer—still occurred, but at only a small fraction of today’s rates. Modern research shows that indoor living amplifies breast cancer risk—especially in the presence of electric lighting—while one to two hours outdoors each day lowers it.
In medieval times, the group most likely to develop breast cancer was nuns, a profession that involved extensive coverage of skin and far more time indoors than the average outdoor laborer. The observation led to centuries of assuming that breast cancer was caused by not bearing children. Current research actually shows little association between childbearing and total breast cancer risk, although breastfeeding might decrease the risk of the estrogen receptor–negative breast cancer subtype. The childbearing hypothesis was further weakened by later findings showing elevated rates of male breast cancer among priests, who lived in similarly indoor, low-sunlight conditions. And as electrification expands across the developing world, breast cancer rates are rising in men as well as women.
There is an important question, however, about the time delay between nighttime light exposure and clinical cancer, and whether changing after-dark lighting habits after diagnosis can impact outcomes. Breast cancer caused by X-rays has an average latency period of 22 years for pre-menopausal women. Shorter latency periods are seen in aggressive cancers in young women. By contrast, longer latencies occur in the less aggressive cancers of older post-menopausal women, for whom most breast cancers—apart from those triggered by hormone therapy—may have been initiated during their reproductive years. Is the same true for cancers associated with evening illumination?
Some U.S. data suggest that might be the case. The 1973 energy crisis, for example, was one of the few times in American history that light pollution departed from its usual steady rise and actually declined. A dip in breast cancer rates appeared in 30–44 year old women exactly 22 years later, in 1995 (see below). For older women, any similar change would have been modest—estimated at around 7%—and impossible to detect because the surge in hormone-therapy–related breast cancers at the time overwhelmed any subtle pattern.
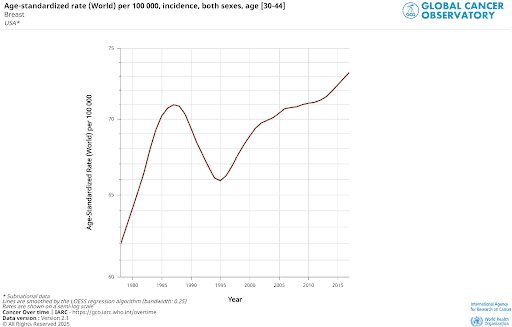
Long-Term Cancer Patterns in Japan and the U.S.
For the long-term impact of light exposure—or lack thereof—on older women, you can look at Japan. Its electrical grid was built at the same time as the United States, but Americans used their power almost exclusively for overhead lighting during the early decades. In contrast, the Japanese traditionally preferred low lights and interior shadow, even as lighting salesmen pressured them to use more electric lighting. Most houses had no wall outlets and just a few overhead sockets for lights, which could be used instead to power appliances. At night, the Japanese covered their windows with amado—opaque storm shutters that effectively blocked light.

Housing rebuilt after WW2 had more wiring infrastructure. By the 1990s, the Japanese adopted compact fluorescent lighting earlier than Americans, and today Japanese living spaces are often brightly illuminated with LED lights. The graph below from Iwase et al. (2024) shows the trend of age at menarche in terms of birth year, so adding roughly 12 years shows when each cohort reached puberty. A mild decline in age of menarche can be seen that reaches its lowest point around the birth year of 1925 and then reverses; these girls would have been reaching age 12 in 1937, as Japan invaded China and home electricity use began to be curtailed.
A renewed and sustained decline appears for girls born after 1932, who would have reached puberty during the late-war and immediate post-war years, when lighting was heavily restricted. For birth cohorts since the mid-1980s, Japanese and American girls have been experiencing menarche at about the same age and, so far, these groups have been getting breast cancer at about the same rates. Earlier, both populations saw substantially lower breast and prostate cancer in Japan.
Looking at the generations for which Japanese and American breast cancer rates were still quite different, Shimizu et al. (1991) found that Japanese women who immigrated to the United States while still young had breast cancer at the same rate as Americans, but women who immigrated when older had a considerably lower rate, even in old age. This indicates that low light exposure during the reproductive years has a long-lasting protective effect for women, and conversely that many breast cancers in older women reflect their earlier light exposure. For men and prostate cancer, however, the researchers found a different story; the cancer rate in Japanese men who immigrated quickly caught up with their American counterparts regardless of age of immigration. The authors hypothesize that this is because latent prostate cancer is extremely common in all populations but only becomes clinically apparent under certain environmental conditions:
“It is well established that the overall and age-specific prevalence of latent prostate cancer has considerably less geographic and racial variation than clinical prostate cancer.”
Thus, with latent cancer already in place, many men can develop clinical disease relatively quickly under the combined effects of aging and electric lighting. These findings have been reinforced by the increase in cancer rates seen in India during the buildout of the electric grid, as mentioned above. So far, the country has seen a 39.1% increase in the age-standardized breast cancer incidence, but a staggering 360% increase in prostate cancer, which likely signals what will happen with breast cancer rates after its longer latency period.

What You Can Change Today
When you manage your light exposure, you lower your risk of clinical prostate cancer at any age, and keeping after-dark light in check during your reproductive years helps protect you from breast cancer down the road. The next question is whether improving your light environment after diagnosis can influence outcomes—or help prevent the emergence of breast cancers that may be silently developing from earlier exposure. This has not been directly investigated. However, one study in Norway found that women diagnosed with breast cancer during the summer months had better outcomes in the south, where UV exposure is higher, than in the north.
During the winter, when latitude-related differences in UV vanish, those survival rates were also indistinguishable. Thus, sunlight after diagnosis appears beneficial, suggesting that avoiding nighttime light might be helpful as well. For those diagnosed during the winter, a sunlamp could be used. Melatonin supplementation is another avenue that has been widely suggested in the literature as a cancer treatment but has not been tested in a large clinical trial—most likely because melatonin offers little patent potential or profit. There is also a deeper issue: for your body, melatonin’s timing is everything. Naturally produced melatonin rises in a wave when darkness falls and then tapers in a precisely timed decline toward morning.
Supplemental melatonin lacks these subtleties and can cause side effects in some people, including headaches, dizziness and hyperglycemia. It may also increase irritability or unusually aggressive behavior. This behavioral shift is tied to timing: melatonin is meant to peak in the middle of the night, when an unexpected disturbance might call for a rapid, protective response. When supplements drive melatonin levels too high during the day, however, the body can misinterpret those signals, leading to misplaced agitation or aggression.
You have many practical ways to support your body’s naturally timed melatonin by reducing blue light and overall brightness at night. One option is using dim orange or red-colored lightbulbs for evening lighting. Most people will be well served by any LED bulb with a color temperature under 2000 K that produces fewer than 350 lumens when the bulb is shielded by a lamp fixture or opaque covering. Go for a dimmer bulb if it isn’t shielded. Having orange electric candles or lanterns that children can carry helps them ease more comfortably into a darkened evening home. Other options include olive oil lamps, beeswax candles safely enclosed in lanterns, orange or red–tinted blue-blocking glasses, and nighttime or dimness settings on phones and other screens.
For overnight light reduction in the bedroom, be mindful of commercial blackout curtains since they often offgas chemicals. Layers of thick cotton curtains can be effective, or you can attach a doubled-over foil space blanket between curtains for a very dark setup. Organic, low–off-gassing blackout curtains are also sold, though usually at a premium. A sleeping eye mask is another simple and effective solution. In addition to reducing your cancer risk, changing your light exposure is also likely to help you feel better and more energetic, as your body realigns with its natural rhythms and receives the full benefits of a healthy circadian cycle.
Putting It All Together
If you or a family member is a cancer patient, you may need to educate your doctor about the connection between light and cancer. Unfortunately, despite the published research and the slow growth in awareness about blue light’s impact on circadian rhythms and sleep, many medical professionals remain unaware of how blue light, melatonin, and hormone regulation intersect with cancer and broader reproductive health. Adding to the problem, medical buildings are often illuminated with the same new, excessively bright blue-heavy LED lights found in other environments.
The IARC’s statement on the probable link between shift work and breast cancer offers a strong starting point for educating others. Explaining how light and hormones are biologically connected—rooted in the way many animals use seasonal light cues to regulate reproduction—helps create a clearer foundation for understanding this relationship. And if you’re not a cancer patient, improving your light environment is a powerful and accessible way to cut your cancer risk, alongside the equally important work of reducing your exposure to chemical pollutants in the air, water or soil. You can eliminate your own light pollution by putting on a pair of blue blockers, changing a light bulb, dimming your phone screen or flipping a switch—simple changes that support the hormone signals your body relies on.
~
Published on December 18, 2025.
If you’ve found value in this article, please share it!
To support the research and health education of AVFC editorial, please consider making a donation today. Thank you.


